Hair restoration procedures require detailed documentation to protect your practice and ensure patient safety. From initial consultations to postoperative care, maintaining accurate records is critical for legal compliance and minimizing risks. Key takeaways include:
- Informed Consent: Clearly document procedure details, risks, benefits, alternatives, and financial terms. Use plain language and ensure patients fully understand before signing.
- Record Retention: Follow state-specific laws for how long to keep medical records (e.g., 7 years in California, 11 years in North Carolina). For minors, retain records until adulthood plus additional years.
- HIPAA Compliance: Safeguard patient health information (PHI) with secure storage and limited access. Respond to patient record requests within required timeframes.
- Photography Standards: Use consistent angles, lighting, and backdrops for clinical photos. Store images securely and ensure patient consent for their use.
- Documentation Phases: Preoperative, intraoperative, and postoperative records should detail every step, including medical history, graft counts, medications, and follow-up care.
Proper documentation isn’t optional - it’s your first line of defense against malpractice claims and regulatory audits. Tools like Prospyr can simplify compliance by integrating patient data, automating workflows, and securing sensitive information.
Legal Requirements for Hair Restoration Records
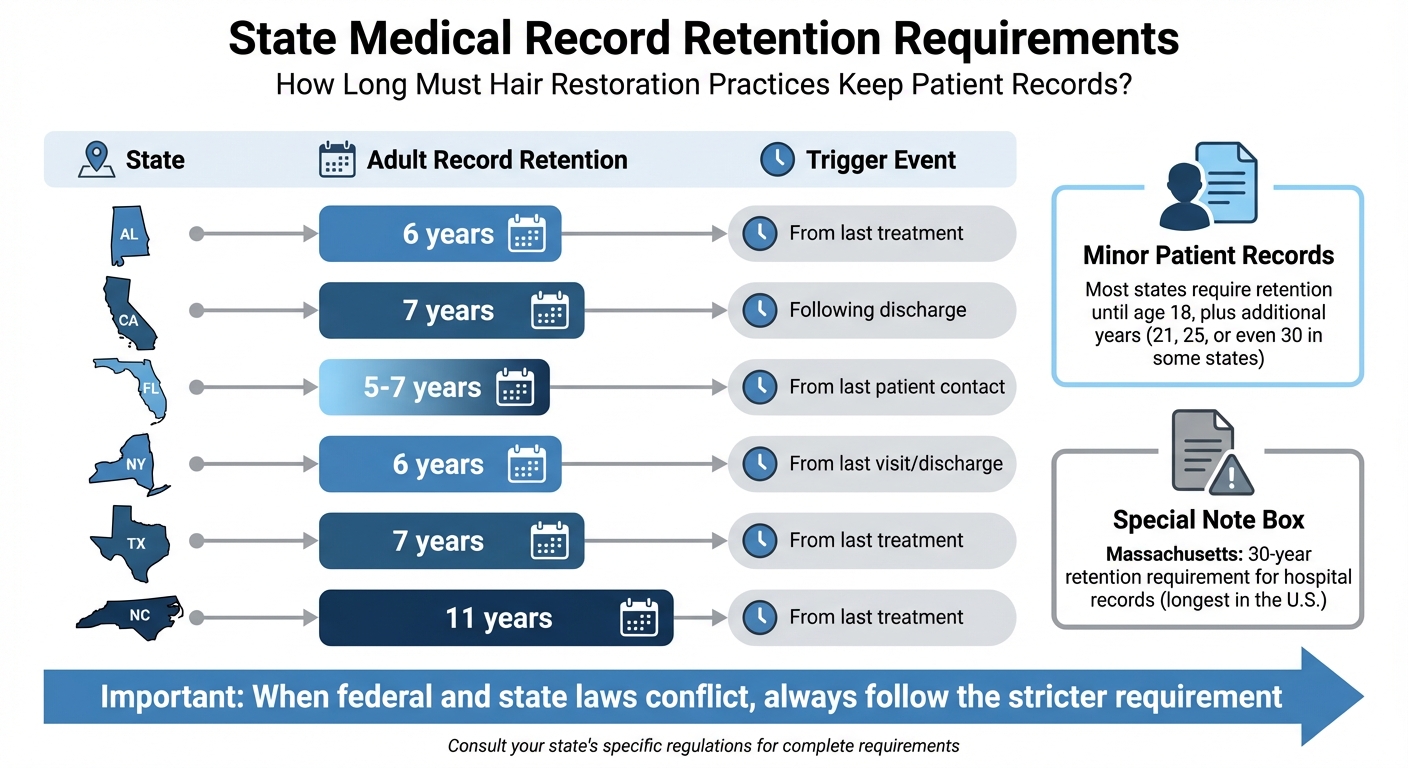
Medical Record Retention Requirements by State for Hair Restoration Practices
Hair restoration practices must navigate a web of federal and state regulations when it comes to creating, storing, and managing patient records through digital intake systems. Staying compliant is not just about following the law - it also protects your practice from potential risks.
Federal Guidelines for Medical Records
The HIPAA Privacy Rule (45 CFR § 164.524) lays out your responsibilities for safeguarding patient health information (PHI). This includes giving patients access to their PHI, such as clinical notes, pre-operative assessments, billing details, and procedure photographs.
While HIPAA requires you to retain administrative records (like policies, training records, and security assessments) for 6 years, it doesn’t specify how long to keep medical records. That’s determined by state laws.
"HIPAA doesn't set retention periods for medical records - state law does." - PatientNotes.ai
If your practice uses or recommends hair restoration topicals classified as cosmetic products, the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) requires keeping adverse event records for 6 years (or 3 years if you qualify as a small business).
State laws further define how long you must retain medical records, creating additional layers of responsibility.
State-Specific Retention Periods
Retention rules for adult patient records vary widely across states, ranging from 5 to 11 years after the last treatment. For example, Florida requires 5 years, California mandates 7 years, and North Carolina has an 11-year requirement.
For minor patients, the rules are stricter. Most states require keeping records until the patient turns 18, plus additional years - sometimes until age 21, 25, or even 30. Massachusetts has one of the longest retention periods, requiring hospitals to keep records for 30 years.
When federal and state laws conflict, follow the stricter requirement. As Kevin Henry from AccountableHQ explains, "When state law provides stronger privacy protections or longer retention than HIPAA, the state law controls".
Here’s a snapshot of retention requirements in different states:
| State | Adult Record Retention | Trigger Event |
|---|---|---|
| Alabama | 6 years | From last treatment |
| California | 7 years | Following discharge |
| Florida | 5–7 years | From last patient contact |
| New York | 6 years | From last visit/discharge |
| Texas | 7 years | From last treatment |
Record Ownership and Patient Access Rights
While you technically own the records, patients have the legal right to access copies. As the custodian of these records, it’s your responsibility to ensure they remain accessible.
Under HIPAA, you must respond to patient access requests within 30 calendar days, with one possible 30-day extension if you notify the patient in writing. However, some states have stricter deadlines. For instance, California and Texas require responses within 15 days, and some states mandate as little as 72 hours.
"The 30 calendar days is an outer limit and covered entities are encouraged to respond as soon as possible." - U.S. Department of Health and Human Services
You can only charge reasonable, cost-based fees for copying records, including labor, supplies (like paper or USB drives), and postage. Charges for searching or retrieving records are not allowed under HIPAA, even if state law permits it.
If you retire or close your practice, you must notify patients and inform them where their records will be stored for the remainder of the retention period. Abandoning records is not an option - you remain responsible for their safekeeping.
sbb-itb-02f5876
Informed Consent Documentation Standards
Informed consent plays a dual role - it ensures patients are well-informed participants in their care and provides legal protection for your practice. In the U.S., failing to secure proper informed consent is considered negligence per se. For elective procedures like hair restoration, where the goal is to enhance quality of life rather than treat a medical issue, the standards for documentation are especially rigorous.
"Informed consent and conflict-free consultation are the moral requirement for any surgery and more so for hair transplant surgery, which is completely elective and is performed not to treat a medical problem but basically improve the patient's quality of life." – Sandeep Suresh Sattur, Department of Plastic Surgery, HAIRREVIVE
With the shift from a paternalistic model of care to one centered on patient autonomy, patients are now active participants in their treatment decisions. To support this, consent forms should be written in plain, accessible language and translated when necessary. Yet, a study revealed that only 26.4% of consent forms included the four required elements - procedure details, risks, benefits, and alternatives - showing a gap in meeting these standards.
Required Components of Informed Consent
An effective consent form should cover the following:
- Diagnosis and Prognosis: Clearly identify the type of hair loss, its expected progression, and the possibility of needing future treatments.
- Procedure Details: Outline the specifics, like the redistribution of existing hair (not creating new hair), the donor area size, and any anesthetics or sedatives involved.
- Risks and Complications: Include potential issues such as infection, scarring, shedding (surgical effluvium), swelling, scabs, and anesthesia reactions. Quantify risks whenever possible to provide clarity.
- Realistic Outcome Expectations: Explain typical results, such as achieving 50% to 70% density in the frontal area and 70% to 90% in the crown. Stress that individual results vary, and a "full head of hair" may not be achievable.
- Treatment Alternatives: Present all options, including medications like Minoxidil or Finasteride, hairpieces, scalp micropigmentation, or even the choice to decline treatment.
- Financial Transparency: Provide a clear breakdown of fees, emphasizing that payment is required regardless of the outcome.
- Administrative Details: Ensure all signatures (patient, physician, witness) are included, along with dates and times. Avoid leaving blank spaces to prevent "blanket consent." For patients who cannot read or write, consider video documentation while adhering to strict privacy measures.
To ensure patients truly understand, use the teach-back method: ask them to explain the procedure and risks in their own words. You can also request a brief handwritten statement confirming they've read the form, had their questions addressed, and understand the risks.
Tailoring consent forms to each phase of treatment is equally important. This ensures the documentation remains accurate as the treatment progresses.
Consent Forms for Different Treatment Phases
Given that hair restoration often involves multiple stages, consent documentation should align with each phase of the process to maintain consistency and clarity.
- Initial Consultation: Provide the consent form during the first meeting, giving patients ample time to review it. Have them sign a receipt to confirm they’ve received the document.
- Day of Surgery: Revisit the consent discussion, ideally in the presence of a relative or witness, before obtaining final signatures. Review details like hairline markings to confirm the patient still agrees to proceed. Ensure all signatures are secured before administering any sedatives or anesthesia, as patients cannot legally consent once sedated. For minors, a legal guardian must sign the consent.
- Subsequent Procedures: For follow-up treatments or touch-ups, don’t rely solely on the original consent form. Use updated, procedure-specific forms to document any changes in the treatment plan, risks, or expectations.
If a patient declines any part of the treatment plan, document their refusal clearly to protect both the patient and the practice legally.
Documentation Standards for Hair Restoration Procedures
Thorough documentation at every stage of hair restoration is essential. It safeguards your practice, ensures patient safety, and provides a reliable record for continuity of care and legal protection. These records build on consent and legal frameworks, aligning the treatment process with best practices.
Preoperative Records
Preoperative documentation lays the groundwork for the procedure. Start with a detailed medical history, noting chronic conditions like heart disease, asthma, diabetes, or clotting disorders. Record any drug allergies, especially to lidocaine, antibiotics, or latex.
Clinical evaluations should include the donor area's suitability and baseline vitals such as blood pressure and pulse. Perform a lignocaine test dose to confirm anesthesia tolerance. Surgical planning must outline the planned hairline design (with photographs), the estimated graft count, and the target scalp area.
Clearly assign roles for each team member, specifying responsibilities like donor harvesting or recipient site preparation. Tasks requiring specialized expertise should be handled by qualified surgeons. Counseling notes should cover expected outcomes, procedure duration, possible future surgeries, and ongoing treatments like minoxidil or finasteride.
Administrative steps include sending a confirmation letter with the procedure date, time, and financial details. Verify patient identity with a photo ID. Preoperative instructions should highlight the importance of stopping aspirin and NSAIDs 10 days before surgery, avoiding alcohol for 7 days, and quitting smoking or nicotine to enhance graft survival. For high-risk patients, such as those with stents or chronic obstructive pulmonary disease (COPD), secure preoperative clearance from their specialists.
Once preoperative records are complete, attention shifts to documenting the surgery itself.
Intraoperative Documentation
During the procedure, keep detailed records of every step. Key elements to document include:
- Vital signs: Heart rate, blood pressure, and oxygen levels at regular intervals.
- Medication log: Note the name, concentration, dose, and timing of all drugs, especially local anesthetics and tumescent solutions.
- Surgical timeline: Record start and end times for anesthesia, harvesting, and implantation phases.
- Graft accountability: Track planned versus actual graft counts, breaking down follicular units by the number of hairs (e.g., 1, 2, 3, or 4 hairs per unit). Provide discharge slips detailing the total units and hairs.
"Intraoperative records should include the vital parameters, drugs administered and their dosage, complications if any and the remedial measures that were carried out, number of grafts planted, the area of scalp covered, the details of the surgical team, and the duration of the surgery with the timings of the beginning and the end of each stage of the surgery." – Venkatram Mysore et al., Hair Transplant Practice Guidelines
Identify all surgical team members by name and role. Specify which tasks were handled by the lead surgeon versus assistants or technicians, ensuring that assistants perform only non-incisional duties like graft implantation into premade slits. Immediately document any complications and the steps taken to address them. Attach intraoperative photos of the hairline design and graft placement for visual evidence.
With the surgery complete, the focus turns to postoperative care and follow-up documentation.
Postoperative Notes
Postoperative documentation begins at the first follow-up, typically on Day 1. Record the condition of the recipient and donor areas, checking for loose grafts, dislodged follicles, or healing issues. Note all medications prescribed or given, such as pain relievers (Tramadol or NSAIDs), corticosteroids for swelling (40–60 mg Prednisone), and topical antibiotics (e.g., Bacitracin or Mupirocin).
Monitor for common postoperative symptoms like swelling, bleeding, itching, or less frequent issues like hiccups. Document the timing of suture or staple removal (usually 7–14 days) and assess wound healing. For FUT procedures, track wound tension to prevent complications like wide scars; for FUE, focus on donor site recovery.
Take standardized outcome photos that match the preoperative ones in angle and lighting. These images are essential for accurate records and potential legal defense. Explain to the patient the expected timeline for graft shedding (2–6 weeks) and regrowth (3–4 months) to set realistic expectations. Confirm that they understand post-op care instructions, including proper hair washing, activity restrictions, and exercise limitations.
Ensure all digital records and photos comply with HIPAA regulations, with written consent for storage and use. Keep medical records for at least five years or longer if legal cases are pending.
Digital Records and Photography Standards
The shift to digital documentation has transformed record-keeping, but it also brings a need for strict compliance with HIPAA regulations, especially for clinical photographs. These images, classified as Protected Health Information (PHI), must be safeguarded just like any other medical record. As discussed earlier, HIPAA violations can result in severe penalties. This makes it essential to establish consistent photography protocols that not only comply with legal requirements but also serve as reliable evidence.
Standardized Photography Protocols
Following standardized photography practices ensures accurate and objective documentation, which is crucial in medical and legal contexts. Maintaining consistency in lighting, angles, and backgrounds helps create records that are both clear and impartial. A study examining civil cases involving surgeons misusing patient photographs revealed that courts ruled partially in favor of the patient in 20 out of 23 cases.
"When a dermatologist takes photographs of the patient at each visit, it provides irrefutable documentation of the skin condition on exact dates and times. These photographs are the best evidence against a medical negligence claim." – Lawrence J. Buckfire, Managing Partner, Buckfire and Buckfire
To achieve consistency, use a controlled setting with neutral backdrops - light blue or gray works well - and ensure proper lighting. Floor markers can help maintain a consistent distance between the patient and the camera. While DSLR cameras are still the top choice, high-resolution smartphones like the iPhone 13 Pro or newer models can be alternatives if used with a tripod and adjusted manually for focus and a 5500K white balance.
Photographic documentation should include standardized views: frontal, vertex, and rear. The frontal view highlights the hairline, the vertex captures the crown pattern, and the rear focuses on donor area density. Additionally, include a patient identification card with their name and ID in at least one photo per series to ensure proper organization and retrieval.
Access to these clinical photographs should be limited to medical personnel directly involved in the patient’s care. If cloud storage is used, ensure a signed Business Associate Agreement (BAA) is in place. For mobile devices, configure them to automatically delete image metadata after uploading to a secure EMR system. Tools like Prospyr offer integrated solutions that simplify these processes, enhancing both security and efficiency in record management.
How Prospyr Supports Record Management
Prospyr consolidates patient data and clinical photographs into a HIPAA-compliant EMR system, ensuring your practice stays aligned with digital security standards. Its digital intake forms streamline patient information collection, including obtaining written consent for photography and storage, reducing the risk of missing critical documentation.
With AI-powered note creation, Prospyr generates detailed records from patient interactions, while integrated task management tools help practices adhere to required retention periods for medical records, including photographs.
Prospyr also enhances security by implementing tiered access controls, restricting sensitive images to authorized personnel. Its cloud-based system uses end-to-end encryption and two-factor authentication to protect against breaches and malpractice risks. As healthcare increasingly adopts cloud services - expected to reach 90% by 2025 - Prospyr equips your practice to meet both current and future compliance demands while streamlining your workflow.
Reducing Compliance Risks Through Proper Documentation
Proper documentation isn't just a formality - it’s a critical layer of protection for your practice. It can shield you from legal trouble, malpractice claims, and regulatory audits. By recognizing common documentation errors and putting systems in place to avoid them, you can significantly reduce compliance risks.
Common Compliance Mistakes
One major pitfall in aesthetic practices is inadequate informed consent. It’s essential to have clear records showing that patients were fully informed about all risks and potential complications before treatment.
Another issue is the lack of documented physician oversight when mid-level practitioners perform procedures. Without this, liability risks increase. Similarly, incomplete patient narratives - missing details about treatment reasons, diagnoses, or full treatment plans - can leave your practice vulnerable during audits or legal reviews. Additionally, HIPAA access violations are a frequent problem. When non-medical staff access more information than necessary, the risk of breaches and penalties rises.
Solutions for Risk Mitigation
To minimize these risks, focus on standardized documentation and controlled access to information:
- Standardized Documentation: Every patient record should include the essentials - history, identifying details, diagnosis, consent forms, prescriptions, referrals, and supporting information. This consistency helps eliminate errors and ensures records are audit-ready.
- Access Controls: Limiting access to sensitive information is key. Prospyr’s EMR system enforces HIPAA’s "minimum necessary" standard by restricting access to authorized medical personnel only. This reduces the chance of breaches and keeps your practice compliant.
Regular staff training is another must. Whether through quick lunch meetings or online quizzes, keeping your team informed about privacy policies and documentation standards is crucial. Prospyr’s task management system can simplify this by automating training reminders and tracking completion, ensuring no one falls behind.
For procedures performed by non-physician providers, documenting physician oversight is non-negotiable. Prospyr’s EMR system makes this easier by automating oversight documentation and restricting sensitive data access to authorized personnel. Plus, its automated workflows prompt staff to fill out all required fields before closing a patient record. This ensures your documentation is as thorough as it needs to be to withstand audits or legal challenges.
Conclusion
Keeping thorough documentation is a cornerstone for both legal protection and high-quality patient care in hair restoration. From initial evaluations to follow-up visits, every record plays a role in upholding care standards and protecting your practice from legal challenges. Medical records must be kept for at least five years, and those related to malpractice claims should be retained until the case is fully resolved.
Since hair restoration is considered an elective procedure, it falls under consumer protection laws. This means patients expect clear, informed consent and open communication. Without proper documentation - like detailed consent forms, operative notes, standardized photos, and witness signatures - your practice could face allegations of negligence. A streamlined system can help you meet these demanding requirements more effectively.
Prospyr's integrated CRM and EMR system offers a solution by combining patient data into a single, HIPAA-compliant platform. It eliminates the risks of fragmented systems by using digital intake forms to standardize patient history and consent collection. Automated workflows ensure all required fields are completed, while task management tools handle training reminders and track their completion. Additionally, access controls safeguard sensitive information, limiting it to authorized personnel. Dr. Daniel Lee of New Life Cosmetic Surgery shared that after switching to Prospyr’s platform, his practice saw a 50% boost in revenue and a 40% increase in appointments.
This unified system not only protects your practice but also supports the high standards discussed throughout this guide. By following these legal guidelines, you can maintain compliance while keeping your practice focused on delivering excellent patient care.
FAQs
What’s the safest way to document informed consent for hair transplants?
The best approach is to use detailed, procedure-specific digital consent forms. These forms should clearly explain the risks, benefits, and alternatives associated with the procedure. To stay compliant with legal standards and help patients fully grasp the information, make it a priority to review and update these forms regularly.
How long should hair restoration records be kept in my state?
The length of time hair restoration medical records must be kept depends on state regulations, which generally require retention for anywhere between 3 to 10 years. However, federal HIPAA regulations mandate that these records be maintained for at least 6 years. To ensure you're following the rules, always verify your state’s specific requirements alongside federal guidelines.
Are pre- and post-op photos considered HIPAA-protected records?
Yes, pre- and post-op photos are considered HIPAA-protected records. This means they fall under strict privacy and confidentiality rules outlined by the Health Insurance Portability and Accountability Act (HIPAA). To use or share these images, you must obtain proper informed consent from the patient. This step ensures compliance with legal and privacy standards, safeguarding the individual's rights and sensitive information.



