Body contouring consent forms are critical for legal and ethical compliance. They protect both patients and providers by ensuring informed decisions and clear communication. Here's a quick summary of what these forms should include:
- Patient Information: Full legal name, date of birth, contact details, and emergency contact.
- Medical History: Current/past conditions, medications, allergies, pregnancy status, and prior surgeries.
- Procedure Details: Clear descriptions of the procedure, risks, benefits, alternatives, and post-care requirements.
- Privacy and Financial Terms: HIPAA compliance, data handling, cost breakdown, and payment policies.
- Signatures: Patient and provider signatures with dates to confirm consent and understanding.
These forms are designed to reduce risks, manage expectations, and ensure transparency. Digital tools like Prospyr can streamline the process, improve accuracy, and enhance record management.
Patient Information Requirements
Getting patient details right is the backbone of legally sound body contouring consent forms. Without accurate information, your practice could face compliance issues or legal risks. A thorough process ensures both regulatory adherence and a safe consent experience.
Patient Identification and Contact Details
Start with the patient’s full legal name - it should match their government-issued ID and medical records exactly. Even small mismatches can lead to documentation errors or legal complications, so always double-check against an official ID.
Date of birth is more than just a formality. It confirms identity, determines the patient’s ability to consent, and dictates how long records must be kept. For minors, this means retaining records for three years after they reach adulthood. For adults, the retention period is typically seven to ten years.
Keep current contact information on file, including address, phone number, and email. Don’t forget an emergency contact - this is crucial for follow-ups or urgent situations. Make it a habit to verify and update these details at every visit. Outdated information can delay communication about complications or recalls, which no one wants.
Assign a unique patient ID through your digital intake system and link it to the consent form. This helps avoid mix-ups and ensures a clear, traceable record. And remember, all patient data must comply with HIPAA Privacy and Security Rules.
Getting these details right is the first step toward properly documenting medical history and procedure-specific information.
sbb-itb-02f5876
Medical History and Screening
Gathering a detailed medical history is a critical step in ensuring patient safety and recognizing potential risks early. This process not only safeguards your patients but also helps protect your practice from avoidable complications.
Medical History Checklist
Your consent form should cover both current and past medical conditions that might influence surgical results. Make sure to ask about conditions like diabetes, heart disease, blood clotting disorders, and autoimmune diseases. It's also essential to document any allergies to anesthesia, latex, or medications used during recovery.
Medication use is another key area to address. Record all prescription drugs, over-the-counter medications, and supplements. For instance, blood thinners can heighten the risk of excessive bleeding during procedures like liposuction or tummy tucks. Don’t overlook pregnancy status for women of childbearing age, as elective body contouring surgeries are typically avoided during pregnancy.
Additionally, note any prior surgeries in the treatment area. For example, previous abdominal surgeries could affect the approach to a tummy tuck. Lifestyle habits, such as smoking and alcohol use, should also be documented since they can interfere with healing.
Digital consent forms can streamline this process by using conditional logic. For example, if a patient indicates they have diabetes, the form can prompt follow-up questions about their management plan and recent HbA1c levels.
Once you’ve gathered a comprehensive medical history, use it to assess whether the patient is a suitable candidate for the procedure they’re considering.
Screening for Procedure Suitability
With accurate patient information in hand, tailor your screening questions to the individual’s medical background and the specific procedure. Not every patient is a candidate for every treatment, so focusing on procedure-specific details ensures you gather the most relevant information.
For surgical procedures like liposuction, arm lifts, thigh lifts, and post-weight loss contouring, ask about recent weight changes, as these can impact tissue quality and skin elasticity. If assessing candidates for a "Mommy Makeover", include questions about past pregnancies and plans for future pregnancies, as these factors can affect both safety and long-term outcomes.
Non-surgical options, such as Kybella, CoolSculpting, and Cellfina, also come with their own set of contraindications. Tailor your screening to identify whether the patient might be better suited for a minimally invasive option instead of surgery. Providing clear pre-consultation instructions can help patients recognize lifestyle or medical factors that could influence their eligibility.
Lastly, ensure all patient data is encrypted and flagged for prompt review if any concerning medical history arises.
Procedure Details and Consent Confirmation
A proper consent form should provide all the necessary details about the procedure, ensuring patients fully understand what they are agreeing to before signing.
Procedure Description
The consent form needs to clearly outline the body contouring procedure. This includes details about the method used, the location where it will take place (hospital, surgical center, or office), the expected timeline for results, and the required post-procedure care. For surgical procedures, results are typically immediate, while non-surgical methods may take weeks or even months to show full effects.
Once the procedure is described, it’s equally important to address risks, benefits, and available alternatives.
Risks, Benefits, and Alternatives
The form should list the specific risks associated with the procedure. For surgical options, these may include infection, scarring, anesthesia complications, blood clots, asymmetry, seromas, hematomas, nerve damage, and tissue loss. Non-surgical procedures carry different risks, such as redness, swelling, hives, soreness, or results that may fall short of expectations, potentially requiring additional treatments.
"By signing it [the consent form], you give permission to the healthcare provider to perform the procedure. You also agree that you understand the risks of the procedure and have realistic expectations." - Cleveland Clinic
The benefits should highlight both aesthetic improvements, like smoother skin and a more defined body shape, and functional advantages, such as improved mobility or relief from issues caused by excess skin. If the procedure addresses functional problems like chronic skin infections or difficulty with daily activities, it’s worth noting that insurance might cover the costs in some cases.
Patients should also be informed about alternative options. Surgical alternatives might include liposuction, tummy tucks, arm lifts, or thigh lifts, while non-surgical options could include treatments like Kybella or CoolSculpting. For cellulite, procedures like Cellfina could be mentioned, and for skin rejuvenation, options such as dermal fillers, microneedling, or chemical peels may be appropriate.
Patient Rights and Acknowledgment
Patients should also be made aware of their rights throughout the process. They must confirm they understand the procedure, its risks, and their rights, including the right to withdraw consent at any point before the procedure begins. The form should require both a signature and a date to legally confirm that the patient has given permission, understands the risks, and has realistic expectations for the results.
For digital consent forms, secure e-signature fields should be included to ensure the signatures are legally valid. These templates can be customized to fit the specific body contouring procedure and the patient’s unique needs. This step complements earlier evaluations of patient history and suitability for the procedure, creating a thorough and transparent consent process.
Privacy and Financial Information
When crafting consent forms for body contouring procedures, it’s not just about the medical details - privacy and financial terms are equally critical. These sections help clarify how patient data will be safeguarded and outline financial obligations, ensuring transparency, legal compliance, and fostering trust between patients and providers.
HIPAA Compliance and Data Privacy
Consent forms should include a HIPAA authorization clause, which gives patients the ability to authorize the use and disclosure of their protected health information (PHI) for treatment, payment, and healthcare operations, as outlined in 45 CFR § 164.506. The document must also detail how patient information will be managed and stored under the HIPAA Security Rule (45 CFR Parts 160 and 164). Additionally, the consent form should address medical record retention requirements, including any state-specific rules that go beyond the general guidelines discussed earlier. This ensures patients are informed about how their records will be handled over time.
Cost and Payment Details
Clear financial terms are essential to avoid confusion or disputes. The consent form should outline the full cost of the procedure, along with any potential extra charges that could arise during or after treatment. Payment terms should be written in straightforward language, specifying whether partial completion of a treatment series leads to proportional charges or if refunds are available. Use bullet points to break down financial obligations for better readability. Additionally, include a checkbox or signature line for patients to confirm they’ve reviewed and understood the payment terms and refund policies. Since body sculpting often involves expensive equipment, the form should highlight any financial disclaimers related to the use of advanced technology or consumables. Proper inventory management ensures these costs are accurately tracked.
Photography and Media Release
Clinical photography serves two purposes: medical documentation and marketing. It’s important to clearly separate these uses. Marketing requires a separate authorization under 45 CFR § 164.508. Patients must have the option to decline the use of their images for promotional purposes without affecting their ability to receive treatment. The form should specify how the images will be used and inform patients of their right to revoke or modify consent by discussing it with their healthcare provider. To make this authorization legally binding, include fields for the patient’s printed name, date, and signature. Finally, all photography and media must comply with regulations like HIPAA, GDPR, and CCPA to ensure the protection of sensitive personal and medical data.
Managing Consent Forms with Prospyr
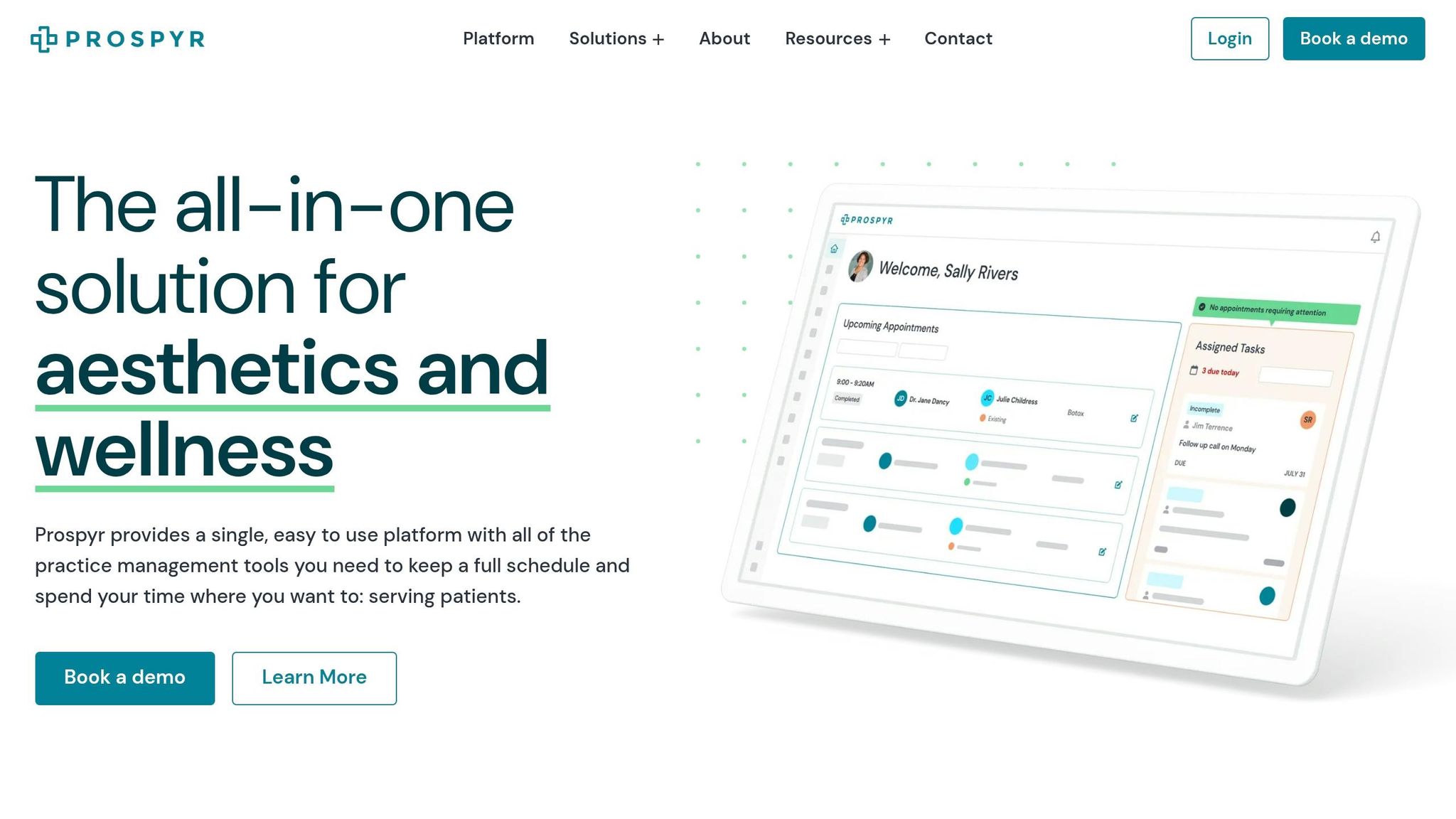
Prospyr makes handling consent forms easier by offering digital tools designed to simplify document management. Their system can cut paperwork time by 50–70% while ensuring full HIPAA compliance. For example, clinics performing around 200 body contouring sessions each month have reported a 25% faster patient flow and no lost forms, thanks to automated backups and seamless integrations.
Customizable Digital Consent Forms
Prospyr's consent forms come with HIPAA-compliant e-signatures and smart conditional logic. Here’s how it works: if a patient selects CoolSculpting, the form will automatically display risks specific to that procedure, such as skin irregularities, while leaving out unrelated surgical warnings. This tailored setup increases form completion rates by 40% and ensures patients only review consents relevant to their treatment.
The forms are mobile-friendly, so patients can fill them out on any device. Automated email and SMS reminders also help reduce no-shows and incomplete forms. This works alongside lead management tools to ensure a smooth transition from inquiry to treatment. With security features like end-to-end encryption, secure cloud storage, and audit logs, Prospyr lowers the risk of data breaches by up to 90% compared to traditional paper systems.
Integration with Practice Management Tools
Prospyr’s digital consent tools integrate seamlessly with practice management systems. Through API connections, it syncs directly with EMR and scheduling systems, automatically pulling patient demographics into consent forms and updating records once a signature is captured. For instance, when a patient signs a body contouring consent, their EMR record updates instantly, and they receive a confirmation SMS - improving operational efficiency by 30%. Practice owners can track these improvements through detailed analytics dashboards.
The system also offers centralized, searchable digital storage complete with timestamps and version history, making compliance audits much easier. Plus, integration with the Media Archive ensures signed consents are stored alongside before-and-after photos, creating a comprehensive patient record in one place.
Compliance Verification

Body Contouring Consent Form Compliance Checklist
Double-checking all elements of a consent form before filing is crucial to protect patients and reduce legal risks. As Forms-Legal emphasizes, "any unconsented touching of a patient constitutes battery, regardless of whether the procedure was performed skillfully". Missing signatures or incomplete disclosures can easily lead to negligence claims.
During this process, ensure that every aspect of the form aligns with regulatory standards. Begin by verifying that patient demographics - such as full name, date of birth, and emergency contact information - are accurately recorded. Pay close attention to the medical history section, focusing on contraindications like isotretinoin use within the past 6–12 months, concurrent medications, or pregnancy status. These details are essential for identifying patients who may not be suitable for the procedure. A thorough review should naturally lead to a final checklist to confirm every required element is in place.
Risk disclosures are another critical component. These must include general risks (e.g., infection, scarring) as well as complications specific to the procedure. For energy-based body contouring treatments, it’s important to document risks related to the patient’s Fitzpatrick skin type, such as the possibility of post-inflammatory hyperpigmentation. Additionally, the form should include a signed acknowledgment that results are not guaranteed and that any before-and-after photos are purely illustrative.
Checklist Review and Staff Verification
The table below provides a quick-reference guide for staff to verify key compliance requirements. It’s essential to confirm that both the patient and the performing provider have signed and dated the form. The provider’s signature serves as proof that the risks were explained verbally as well as in writing. Also, ensure the form includes a signed HIPAA PHI authorization clause.
| Consent Form Component | Compliance Requirement | Staff Verification Check |
|---|---|---|
| Patient Demographics | Full name, date of birth, and contact details | [ ] Verified |
| Procedure Specifics | Device/product brand names and treatment areas | [ ] Verified |
| Medical Screening | Medications, allergies, and pregnancy status | [ ] Verified |
| Risk Disclosure | Inclusion of general and procedure-specific risks | [ ] Verified |
| Alternatives | Discussion of other options or no treatment | [ ] Verified |
| Results Disclaimer | Acknowledgment that outcomes vary and aren't guaranteed | [ ] Verified |
| HIPAA/Privacy | Authorization for PHI use and media release | [ ] Verified |
| Signatures | Dated signatures from both patient and provider | [ ] Verified |
Once the form is finalized, it must be stored securely in a system that complies with retention and privacy regulations. Whether using a digital or physical filing system, make sure completed forms are both secure and easily accessible for audits or reviews. This step is critical for maintaining compliance and ensuring patient trust.
FAQs
What makes a consent form legally valid?
A consent form is considered legally valid when it clearly outlines how patient information or images will be used, secures the patient’s voluntary agreement, and complies with HIPAA regulations. This includes obtaining proper signatures, implementing security measures to protect the data, and providing detailed disclosures about how the information will be used and the patient’s right to revoke consent.
When should a patient sign the consent form?
Patients are required to sign the consent form prior to the body contouring procedure. This step confirms they have agreed to the treatment and understand the details, fulfilling both legal and regulatory obligations.
How long must consent forms be kept in the U.S.?
In the United States, HIPAA regulations require that consent forms be retained for a minimum of 6 years. This policy is in place to ensure compliance with federal guidelines and maintain proper record-keeping practices.


